Quiz Summary
0 of 3 questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 3 questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
-
 Canadian Society of Pathology
Canadian Society of Pathology
Case Of The Month
Case and discussion contributed by:
Louis-André Julien M.D. M.Sc. FRCPC, René P. Michel, MD,CM FRCPC. and Manon Auger MD,CM FRCPCDepartment of Pathology
McGill University
Montreal, Canada52-year-old woman with idiopathic unilateral effusion around a breast implant, 10 years after implantation for aesthetic purposes.
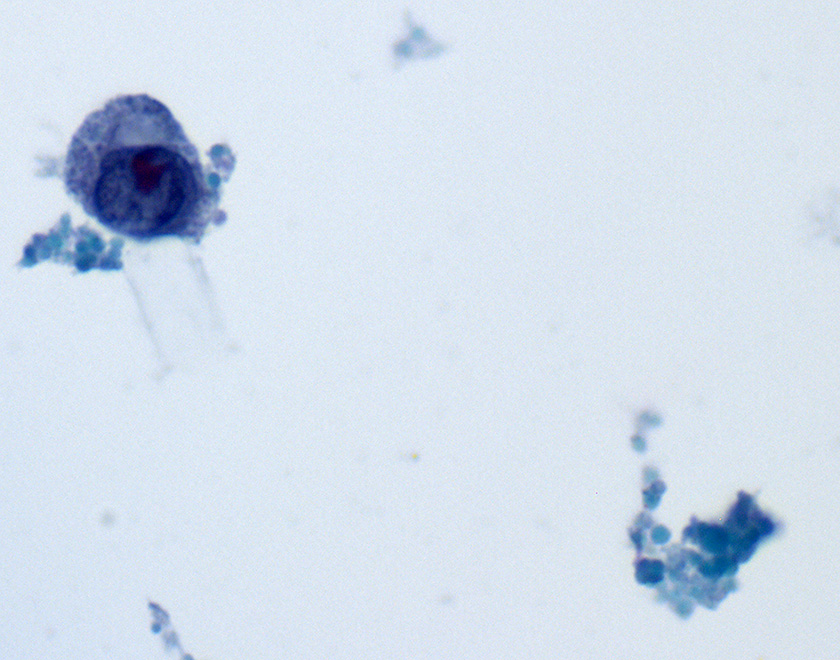
Direct smear – Pap stained, 600X 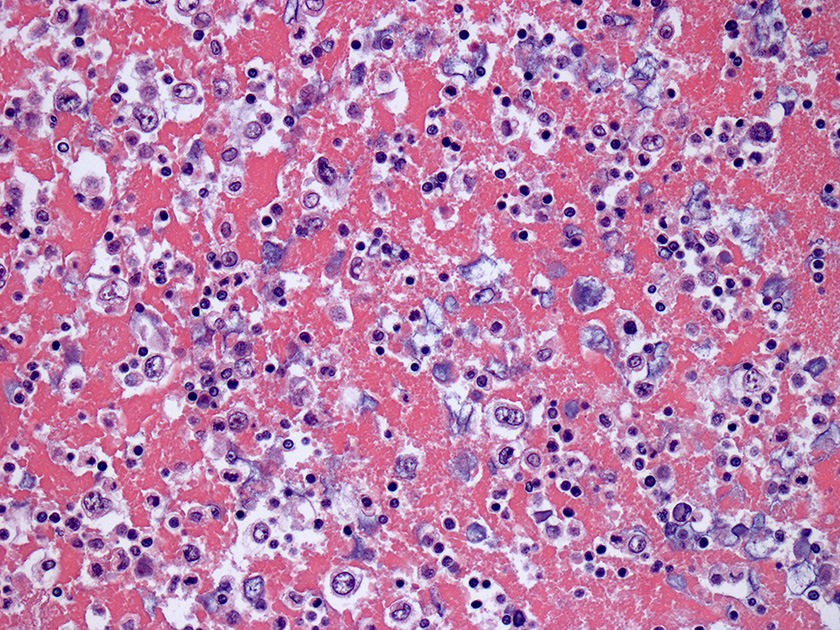
Cell block – HE stained, 200X 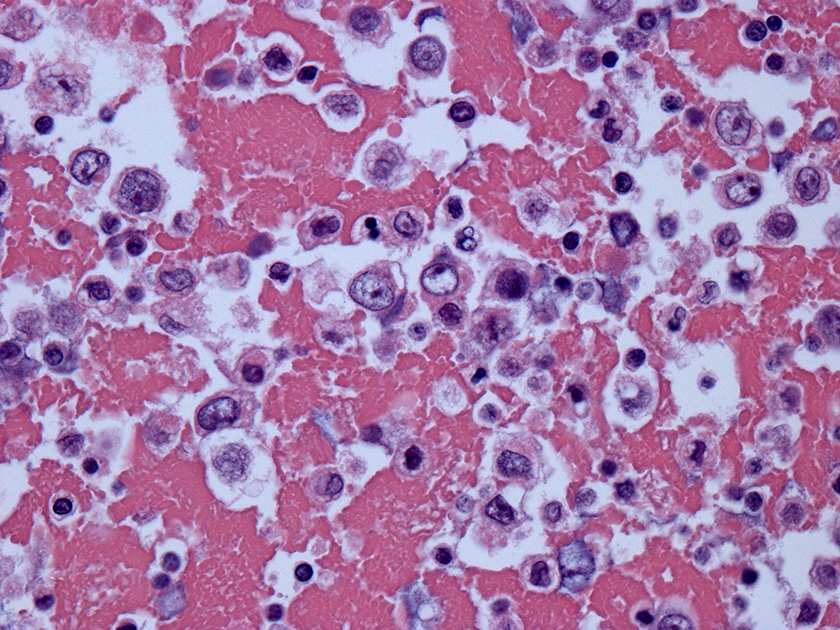
Cell block – HE stained, 400X 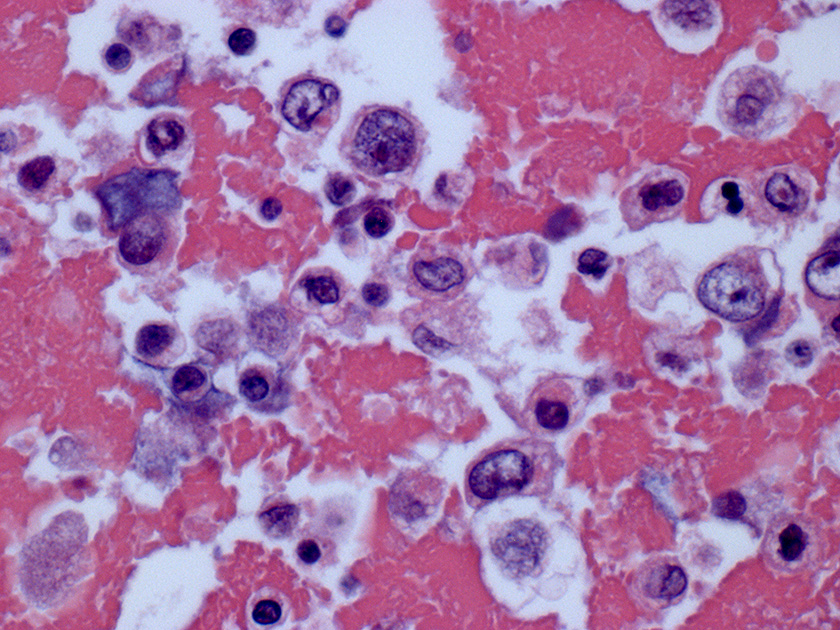
Cell block – HE stained, 600X
Discussion
Given the clinical presentation and cytomorphological findings shown, the diagnosis of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) must be considered and is the diagnosis in this case.
Early postoperative breast implant associated effusions, defined as occurring less than a year after implantation, are often caused by surgical or implant-related complications and are typically unrelated to BIA-ALCL.1 In contrast, delayed effusions are rare and occur more than a year after surgery in about 0.1 to 0.2% of patients.1 About 90% of these are reactive effusions, commonly related to infection, trauma or rupture of implant, but a number remain idiopathic and must be considered suspicious for BIA-ALCL.1-3 The incidence of BIA-ALCL is estimated at 1 per 0.5 – 3 millions of women with implants per year with a recently reported relative risk of 421.8, highly suggestive of a direct or indirect causal role of the breast implants, but the absolute cumulative risk remains small and estimated at 29 per million at age 50 and 82 per million at age 70.4,5 BIA-ALCL represents about 9% of delayed breast-implant associated seromas as published recently in a case series and occur on average 10 years after implantation, yet the interval between surgery and effusions caused by BIA-ALCL varies greatly, with rare cases occurring less than a year after implantation or up to 39 years later. 2,6,7 BIA-ALCL is a T-cell neoplasm that is usually considered an indolent disease frequently presenting with a spontaneous late onset effusion around a breast implant with overall survival rates of 93% and 89% at 3 and 5 years respectively, but some patients present with a palpable mass or lymphadenopathy, which portends poorer prognosis.8-11
Cytologic evaluation of breast implant associated effusions is considered the gold standard for diagnosis of BIA-ALCL. Morphological appearance on smears is typically that of a highly cellular specimen with a fibrinous, granular or necrotic background. Neoplastic lymphocytes are typically 1.5 to 5-fold larger than mature lymphocytes, have abundant vacuolated cytoplasm and pleomorphic irregular nuclei with prominent nucleoli, including some kidney-shaped or horseshoe-shaped nuclei (“Hallmark cells”), multinucleated cells or Hodgkin-like cells.2,12 These neoplastic cells most commonly represent the predominant cellular population, admixed with variable numbers of small reactive lymphocytes and macrophages. By immunohistochemistry, CD30 is positive in all cases of BIA-ALCL, being strong and uniform in most neoplastic lymphocytes, and ALK is always negative.4 Loss of several T-cell antigens including CD3, CD5, CD2 or CD7 is also very common, but the majority of cases are positive for CD4, retain expression of CD43 and often express cytotoxic molecules. 6,12 Immunophenotyping by flow cytometry on fresh samples as well as molecular analyses for TCR gene rearrangement on the cell block may aid in confirming the diagnosis. However, identification of the neoplastic cells of BIA-ALCL by flow cytometry is challenging, potentially leading to false negative results, and requires availability of T-cell panel antibodies and expertise in gating to arrive at a precise diagnosis.12-14 Therefore, and as per recent NCCN guidelines, it is good to keep in mind that larger volumes of fluid yield more accurate diagnosis; if possible, obtain more than 50mL for cytology and cell block, and more than 10mL for immunophenotyping by flow cytometry where this expertise is available.12,15
In the presence of anaplastic large cell lymphoma cytomorphology, a crucial differential diagnosis consideration is the possibility of a non-breast implant-associated ALCL involving the breast, particularly systemic ALCL, ALK-negative or ALK-positive, as these are aggressive lymphomas often presenting with advanced and high stage disease with multiple lymphadenopathy and rapid progression without treatments, but also the possibility of primary cutaneous ALCL that conversely have a very favorable prognosis.4,16 Therefore, correlation with clinical and imaging parameters as well as history of breast implant and ALK status (expression or rearrangement) is always necessary for an accurate diagnosis.
The differential diagnosis should also include other subtypes of B-cell or T-cell lymphomas with large cell or anaplastic features, particularly if there is a history of systemic lymphoma. In fact, the vast majority of lymphomas involving the breast, either primarily or secondarily in the context of systemic disease, are B-cell lymphomas, diffuse large B-cell lymphoma and extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) in particular, whereas lymphomas other than BIA-ALCL occurring in the breast specifically in association with breast implants are exceedingly rare.5,17 Importantly, CD30 per se, irrespective of cytomorphology, is not specific for BIA-ALCL as it can be expressed in other lymphomas and to some extent in a variety of benign conditions.2,12 Possibility of non-lymphoid malignancies involving the breast, particularly primary breast carcinoma, that is much more common in the breast than BIA-ALCL, but also metastatic carcinoma, melanoma or other poorly differentiated neoplasms should also be considered. Although reactive effusions are much more common than BIA-ALCL, the predominant inflammatory cell type in such effusions varies widely and large reactive macrophages with nuclear indentations may be found and must be distinguished from the neoplastic cells of BIA-ALCL.3,12 Cytological findings, immunohistochemistry and correlation with clinical information should resolve these differential diagnoses in most of these instances.
References
- Clemens MW, Brody GS, Mahabir RC, Miranda RN. How to Diagnose and Treat Breast Implant-Associated Anaplastic Large Cell Lymphoma. Plast Reconstr Surg. 2018;141:586e-599e.
- Di Napoli A, Pepe G, Giarnieri E, et al. Cytological diagnostic features of late breast implant seromas: From reactive to anaplastic large cell lymphoma. PLoS ONE. 2017;12:e0181097.
- Di Napoli A. Achieving Reliable Diagnosis in Late Breast Implant Seromas: From Reactive to Anaplastic Large Cell Lymphoma. Plast Reconstr Surg. 2019;143(suppl 3):15S-22S.
- Feldman AL, Harris NL, Stein H, et al. Breast implant-associated anaplastic large cell lymphoma. In: Swerdlow SH, Campo E, Harris NL, et al, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. Revised 4th ed. International Agency of Research on Cancer. 2017:421-422.
- de Boer M, van Leeuwen FE, Hauptmann M, et al. Breast Implants and the Risk of Anaplastic Large-Cell Lymphoma in the Breast. JAMA Oncol. 2018;4(3):335-341.
- Barbe E, de Boer M, de Jong D. A practical cytological approach to the diagnosis of breast-implant associated anaplastic large cell lymphoma. 2019;30:363-369.
- Leberfinger AN, Behar BJ, Williams NC, et al. Breast Implant-Associated Anaplastic Large Cell Lymphoma: A Systematic Review. JAMA Surg. 2017;152:1161-1168.
- Clemens MW, Jacobsen ED, Horwitz SM. 2019 NCCN Consensus Guidelines on the Diagnosis and Treatment of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). Aesthet Surg J. 2019;39(suppl 1):S3-S13.
- Clemens MW, Nava MB, Rocco N, Miranda RN. Understanding rare adverse sequelae of breast implants: anaplastic large-cell lymphoma, late seromas, and double capsules. Gland surg. 2017;6:169-184.
- Clemens MW, Medeiros LJ, Butler CE, et al. Complete Surgical Excision Is Essential for the Management of Patients With Breast Implant-Associated Anaplastic Large-Cell Lymphoma. J Clin Oncol. 2016;34:160-168.
- Laurent C, Delas A, Gaulard P, et al. Breast implant-associated anaplastic large cell lymphoma: two distinct clinicopathological variants with different outcomes. Ann Oncol. 2016;27:306-314.
- Julien LA, Michel RP, Auger M. Breast implant-associated anaplastic large cell lymphoma and effusions: A review with emphasis on the role of cytopathology. Cancer Cytopathol.
- Kesler MV, Paranjape GS, Asplund SL, McKenna RW, Jamal S, Kroft SH. Anaplastic large cell lymphoma: a flow cytometric analysis of 29 cases. Am J Clin Pathol. 2007;128:314-322.
- Muzzafar T, Wei EX, Lin P, Medeiros LJ, Jorgensen JL. Flow cytometric immunophenotyping of anaplastic large cell lymphoma. Arch Pathol Lab Med. 2009;133:49-56.
- National Comprehensive Cancer Network. National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology, T-cell lymphomas, Version 1.2020. Published January 6, 2020. Accessed April 2020. https://www.nccn.org/professionals/physician_gls/pdf/t-cell.pdf.
- Savage KJ, Harris NL, Vose JM, et al. ALK- anaplastic large-cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: report from the International Peripheral T-Cell Lymphoma Project. 2008;111:5496-5504.
- 1
- 2
- 3
- Current
- Review
- Answered
- Incorrect
-
Question 1 of 3
1. Question
What is the most likely diagnosis considering the clinical history and cytomorphology?
CorrectIncorrect -
Question 2 of 3
2. Question
What immunophenotype would be most consistent with the diagnosis?
CorrectIncorrect -
Question 3 of 3
3. Question
Considering the epidemiology of this condition, which of these statements is true?
CorrectIncorrect
