Quiz Summary
0 of 3 questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 3 questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
-
 Canadian Society of Cytopathology
Canadian Society of Cytopathology
Case of the Month
Case and discussion contributed by:
Marc Pusztaszeri, MD
Associate Professor
Department of Pathology
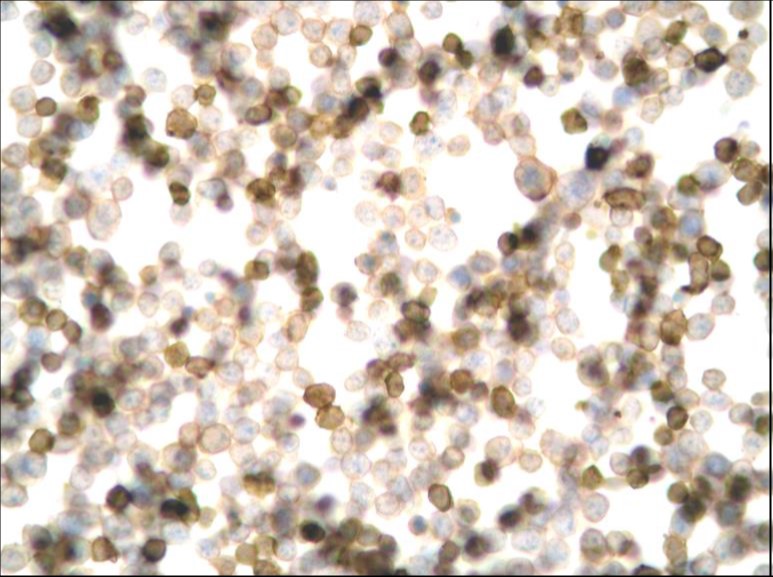
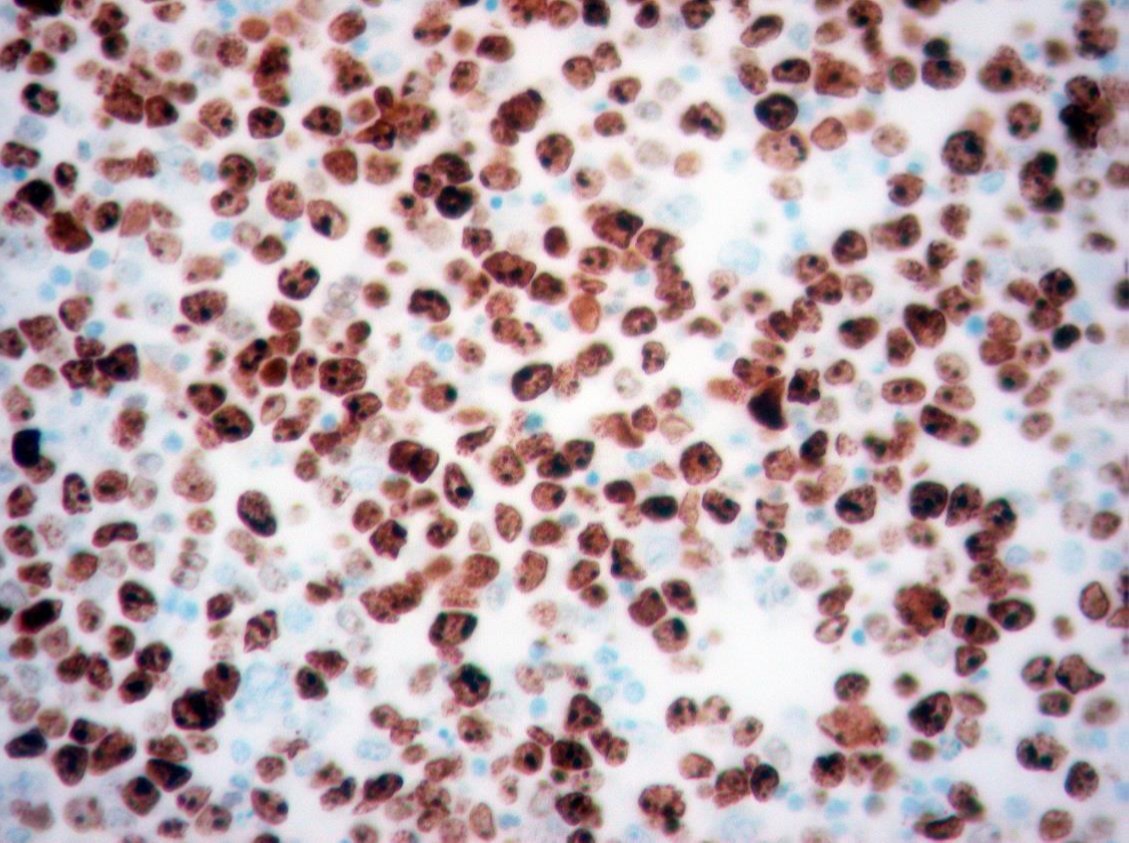
McGill UniversityCD45 IHC
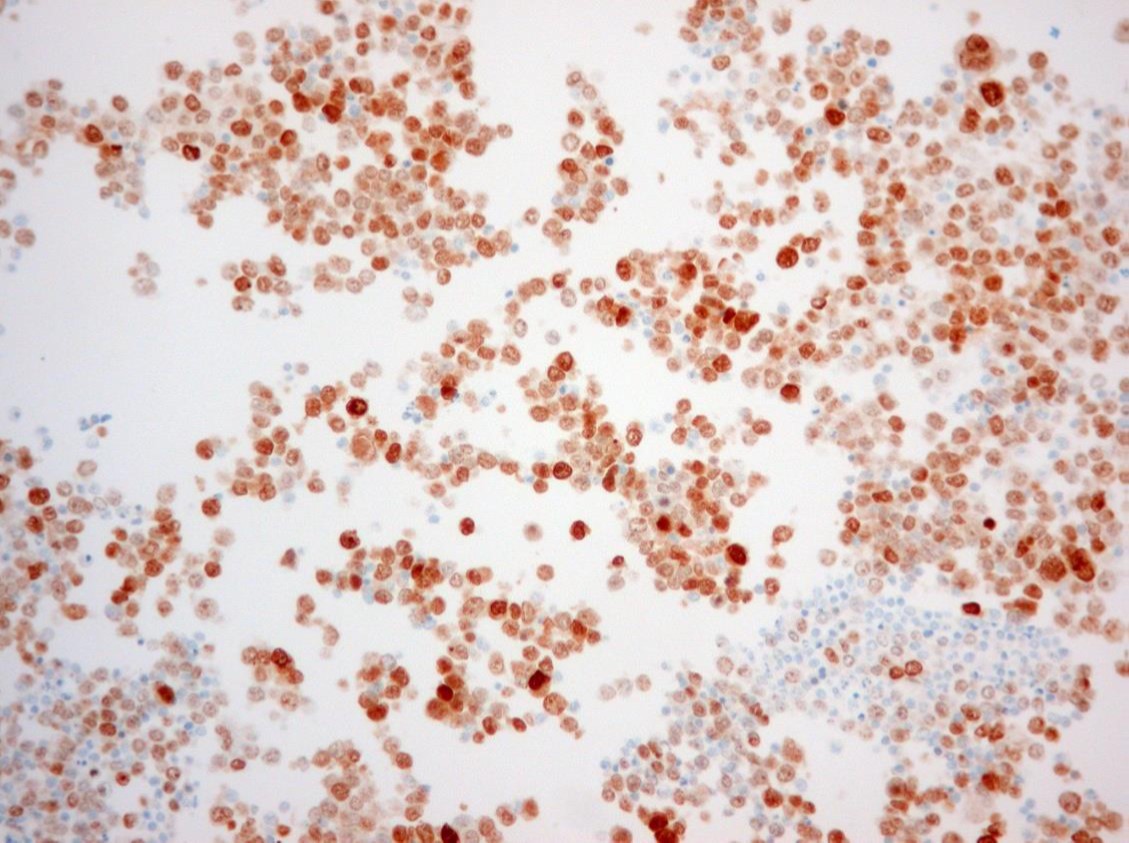
MUM-1 IHC
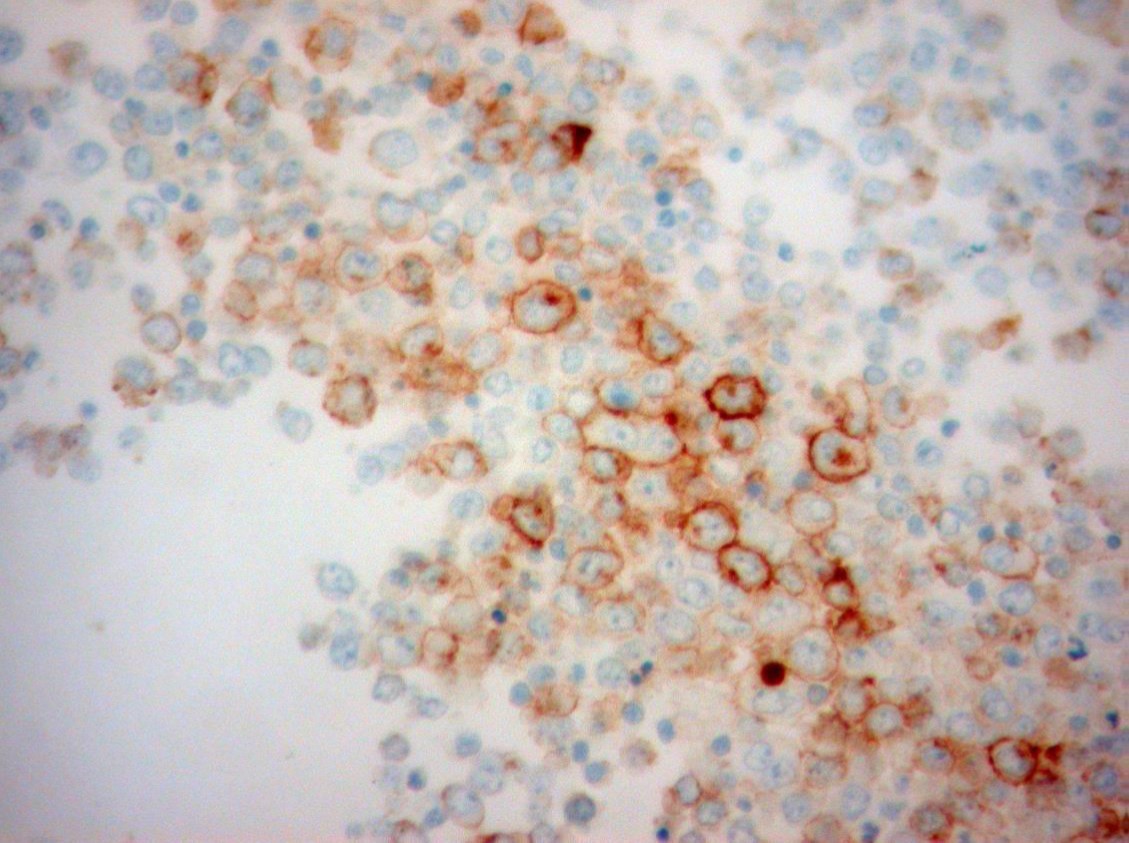
CD30 IHC
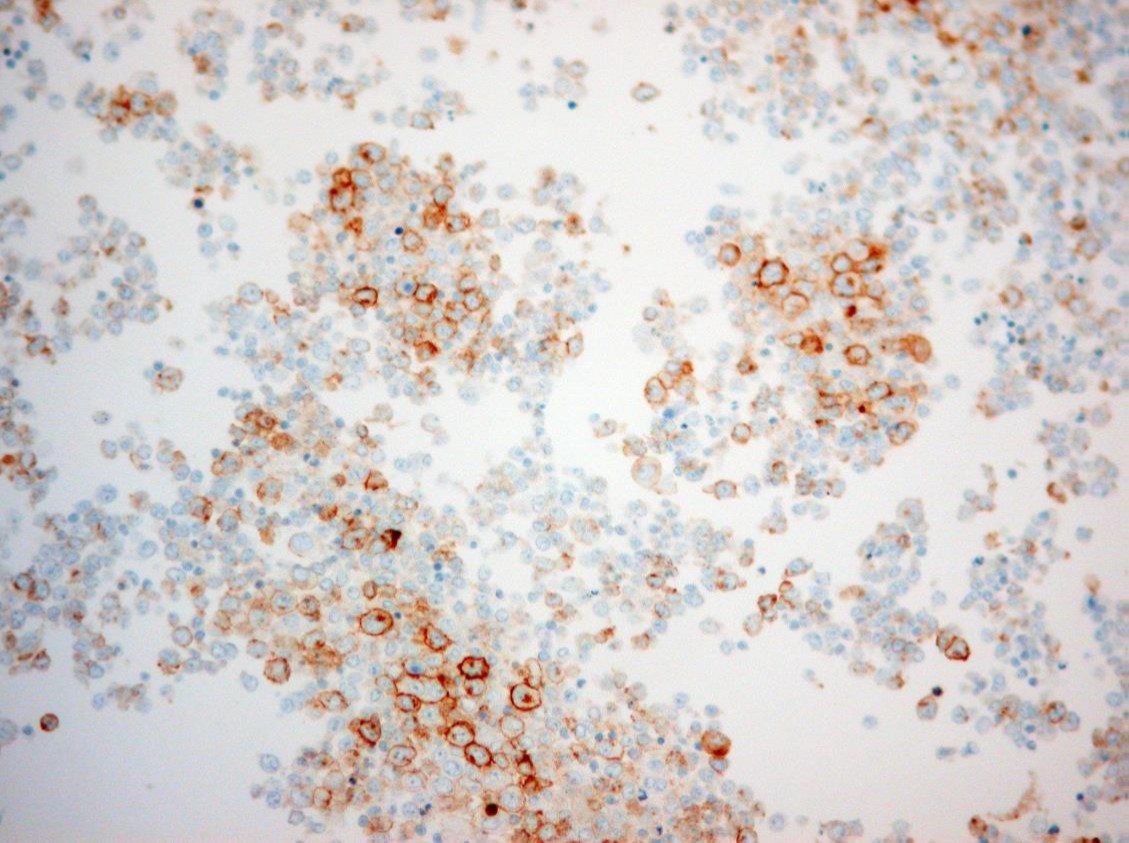
CD138 IHC
HHV-8 IHC
EBV-EBER
Discussion:
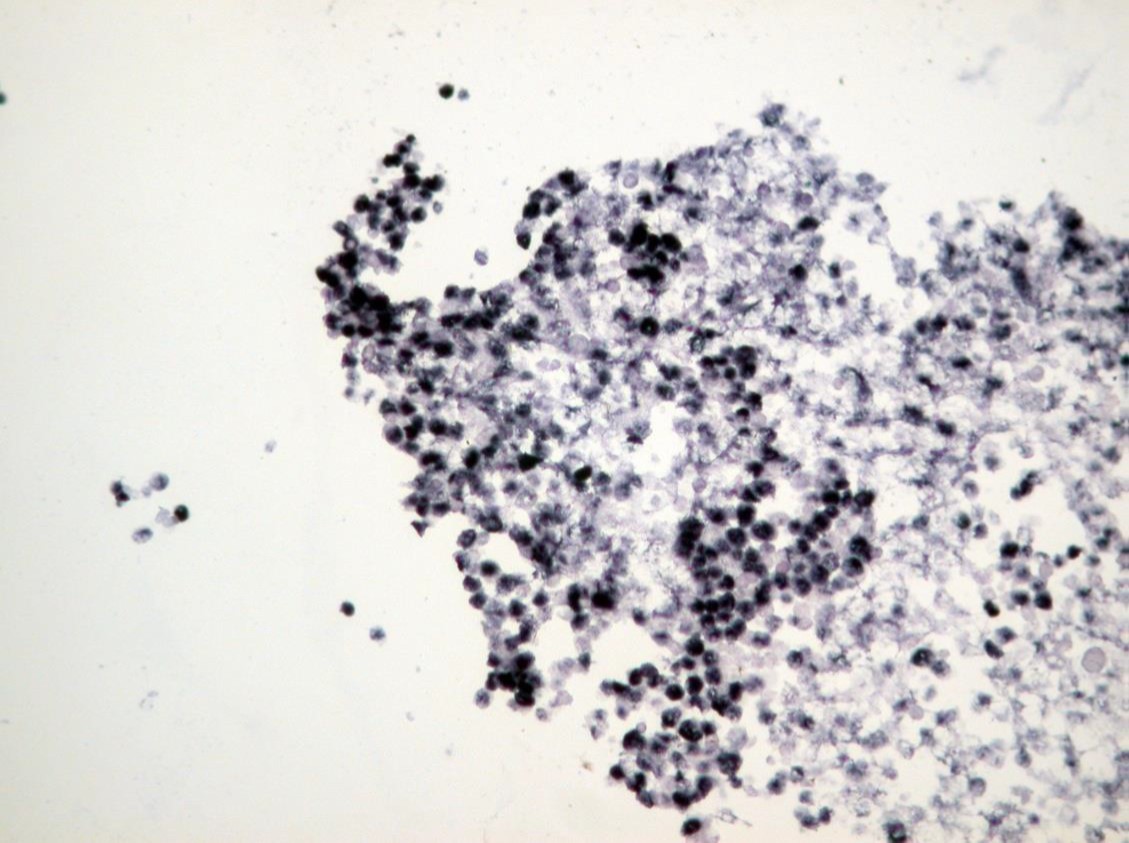
The pleural fluid specimen was very cellular, composed of discohesive malignant cells characterized by high nuclear-cytoplasmic ratio, irregular nuclear membrane, coarse chromatin and prominent nucleolus. There were abundant mitotic and apoptotic figures. By immunohistochemistry (IHC), the tumor cells showed strong expression of MUM-1/IRF-4, focal expression of CD30 and CD3, and weak expression of CD138. There was no expression of CD45 (LCA) and CD20. Proliferative index (MIB-1/Ki-67) was high (>70%). Co-infection with HHV-8 and EBV was detected by IHC and by in situ hybridization (EBER), respectively. Diagnosis of primary effusion lymphoma was established, although initial flow cytometry evaluation did not show evidence of B or T cell clonality. However, after cytological correlation, additional markers were tested including CD38, Ki-67 (MIB-1) and MUM-1(IRF-4), allowing identification of an abnormal population.
Primary effusion (body cavity–based) lymphoma (PEL) is a rare subtype of diffuse large B-cell lymphoma (DLBL) universally associated with human herpes virus 8 (HHV-8, also known as Kaposi sarcoma herpes virus (KSHV)) that manifests with a pleural, pericardial, or peritoneal effusion, usually without an associated mass lesion, lymphadenopathy, or organomegaly (1-3). Therefore, the diagnosis of PEL rests on the cytologic examination of body fluid. Rare cases of ‘extracavitary’ or ‘solid’ PEL that either followed or preceded body cavity involvement or were the only site of involvement in some cases have been described (2). Most cases (about 75%) of PEL arise in the setting of HIV infection; rare cases arising in transplant recipients or elderly patients with secondary immunodeficiencies have been reported. The pleural effusion may the first manifestation of AIDS‐related diseases (2). The cytologic diagnosis of malignancy is usually straightforward. The cells are dispersed and always large, ranging from plasmablastic/immunoblastic (round nucleus with prominent nucleolus) to anaplastic (irregular, multilobate nucleus) in morphology. They have round or irregular nucleus, coarse chromatin, prominent nucleolus, and abundant basophilic cytoplasm (Romanowsky/MGG type of stain). Mitoses and apoptotic bodies are usually present and can be numerous. On IHC, most cases have a “null-cell immunophenotype” (CD20, PAX5, and CD3 negative; CD138, CD30, CD45 positive) and rare cases have a T-cell immunophenotype (2-4). Depending on the antibody panel selection, flow cytometry may fail to identify a clonal population in PEL. Therefore, a correlation with the cytomorphology and immunophenotype on the Cellblock material is essential. Although cases of PEL generally have a null‐cell immunophenotype, they are considered to be of B‐cell derivation on the basis of clonal rearrangement of the heavy immunoglobulin (Ig) genes and recent polymerase chain reaction (PCR)‐based findings of a preferential expression of certain lambda light chain genes, suggesting clonal proliferation by an antigen selection process (2). Importantly, all cases are positive for HHV-8 and most cases (80%) are also positive for Epstein Barr virus (EBV) (1-4). The prognosis is extremely poor with a median survival of < 6 months (2-4). PEL is generally resistant to chemotherapy, however, recent advances exploring mechanisms of HHV-8 oncogenesis have suggested potential novel therapeutic targets.
The differential diagnosis of PEL includes the following entities: DLBL other than PEL, pyothoraxassociated lymphoma, anaplastic large cell lymphoma, post-transplant ymphoproliferative disorder, carcinoma, melanoma (2,4). PEL is morphologically similar to the immunoblastic type of DLBL. If the patient has a mass lesion, lymphadenopathy, or organomegaly along with the effusion, the lymphoma is not classified as a PEL but rather as a conventional DLBL. IHC can also distinguish these entities, because DLBL expresses B-cell markers and is negative for HHV-8 (2,4). Pyothorax-associated lymphoma (PAL) is a rare, EBV-associated subtype of DLBL that develops after longstanding chronic pleural inflammation, and there is usually an associated pleural mass (3,4). Like other DLBLs, it expresses B-cell markers and is negative for HHV-8. Anaplastic large cell lymphoma (ALCL) sometimes manifests with a pleural effusion. The “hallmark cells” (with horseshoe-shaped nuclei) typical of ALCL can be seen in some cases of PEL, but ALCL is negative for HHV-8 (2,4). In transplant recipients, the possibility of a post-transplant lymphoproliferative disorder (PTLD) should be considered. PTLDs are associated with EBV but are negative for HHV-8. Finally, carcinomas and melanoma can be excluded by the absence of immunoreactivity for keratins and melanocytic markers (S-100 protein, SOX-10 and HMB-45). In conclusion, diagnosis of PEL should be considered in cases of malignant lymphomatous effusion in a proper clinical setting (effusion without tumor mass, in HIV positive, immunosuppressed or elderly patient). IHC is essential to confirm the diagnosis (HHV-8 detection is required (confirmatory test)).
References:
1) Patel S, Xiao P. Primary effusion lymphoma. Arch Pathol Lab Med. 2013;137:1152-4.
2) Brimo F, Michel RP, Khetani K, Auger M. Primary effusion lymphoma: a series of 4 cases and review of the literature with emphasis on cytomorphologic and immunocytochemical differential diagnosis. Cancer. 2007;111(4):224-33.
3) Swerdlow SH (eds). WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th Edition 2008, IARC, Lyon. France.
4) Cibas ES, Ducatman BS. Cytology Diagnostic Principles and Clinical Correlates. Saunders; 4th edition, 2014.
- 1
- 2
- 3
- Current
- Review
- Answered
- Incorrect
-
Question 1 of 3
1. Question
The MOST LIKELY diagnosis in the clinical context is:
CorrectIncorrect -
Question 2 of 3
2. Question
Which immunohistochemical (IHC) stain is expected to be positive in this condition?
CorrectIncorrect -
Question 3 of 3
3. Question
Which of the following statement regarding this condition is true?
CorrectIncorrect